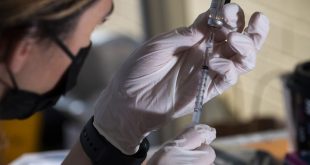
Pfizer and BioNTech announced last month that data from a trial of their COVID-19 vaccine in children found that it provided strong protection in kids ages 5 to 11. The companies have submitted the data to federal regulators for review, and said they plan to submit a formal request for an emergency-use authorization “in the coming weeks.” The Food and Drug Administration …
Read More »Have a kid from 5 to 11 who will get a COVID-19 shot once they’re approved? We want to hear from you
October 6, 2021
 Jago Times "English Edition" Best Online Newspaper
Jago Times "English Edition" Best Online Newspaper